Chest Pain
R/O Acute Myocardial Infarction
The Common Vein Copyright 2008
Daniela Budiu MD Ashley Davidoff MD
Background
Chest pain is a common clinical presentation and its cause can range from a clinically benign musculoskeletal entity to a potentially life threatening situation. In this module we explore the approach to the patient with chest pain in whom we suspect that the cause is cardiac in origin based on initial clinical history and examination. The “classical” patient may be a hypertensive diabetic who presents to emergency department with severe left sided chest pain – pressure like, for 2 hours. The pain could be associated with lightheadedness, palpitations and shortness of breath. Radiation to the jaw and left arm is another classical feature. Although angina may respond to nitroglycerin, the pain of acute myocardial infarction is not relieved. Acute coronary syndrome is a term that describes this clinical scenario, and confirmatory diagnosis requires a 12 lead EKG, blood tests and close attention to vital signs.
At this point, it is important to distinguish between unstable angina and myocardial infarction. Two basic EKG forms are recognized; non ST segment elevation myocardial infarction (NSTEMI) and ST segment myocardial infarction (STEMI).
Terminology
Acute coronary Syndrome
Acute coronary syndrome (ACS) is the term used to describe any one of the 3 following entities: unstable angina, non-ST elevation myocardial infarction (non Q wave MI) and ST elevation myocardial infarction (Q wave MI). Myocardial ischemia is the pathophysiological substrate for all the entities above, encompassed by the term acute coronary syndrome. Myocardial ischemia is caused by an imbalance between myocardial oxygen demand and supply secondary to coronary stenosis, obstruction, thrombosis or spasm. The chest pain in an acute coronary syndrome is caused by ischemia.
Unstable angina
Unstable angina is the clinical presentation of acute myocardial ischemia characterized by new onset chest pain within 1 month, chest pain at rest, chest pain lasting more than 20 minutes and not improving with nitroglycerin or chest pain more severe, more frequent and lasting longer than previously, without evidence of myocardial necrosis (no elevation of cardiac enzymes) and with or without EKG changes of ischemia. UA is a clinical diagnosis.
Myocardial infarction
Myocardial infarction is a circulatory disorder that is characterized by the necrosis of myocardial tissue caused by myocardial ischemia, and clinically characterized by the presence of symptoms of acute myocardial ischemia (ischemic chest pain or chest pain equivalent as dyspnea, diaphoresis, lightheadedness, palpitations), associated with characteristic EKG changes and biochemical markers of myocardial necrosis as reflected by typical rise and fall of cardiac enzymes: troponin I or T, CK-MB. Pathological diagnosis of myocardial infarction requires evidence of myocyte cell death (i.e. necrosis of the myocardium) as a consequence of prolonged ischemia.¹
An alternate approach to classifying these entities is based on the EKG, depending on the presence of elevation of the ST segment
Non ST elevation:
unstable angina (UA)
myocardial infarction (NSTEMI)
The incidence of finding thrombus in the coronary arteries in these patients is between 35-70%.
ST elevation:
ST elevation myocardial infarction (STEMI)
The incidence of finding thrombus in the coronary arteries in these patients is about 90%.
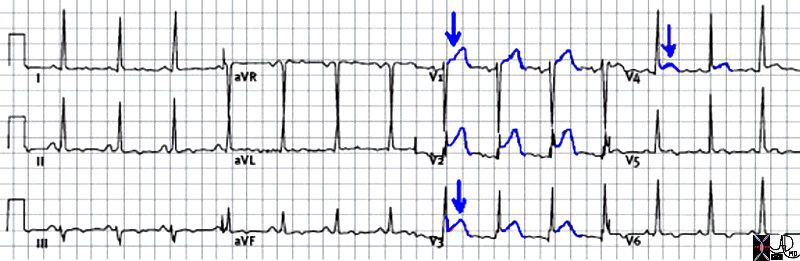
ST elevation in Anterior STEMI |
| The 12 lead EKG shows ST segment elevation in leads V1- V4 (blue) indicating a ST segment elevation myocardial infarction. (STEMI )
76096 heart cardiac infarction STEMI 12 lead EKG blue lead shows ST elevation in leads V1 -V6 consistent with STEMI EKG Courtesy Philips Medical Systems |
The obstruction in UA/NSTEMI is the result of one of 4 main pathophysiologic processes: 1. plaque rupture with superimposed nonocclusive thrombus (the most common cause), 2. dynamic obstruction such as coronary spasm, 3. progression of a mechanical obstruction ( advancing atherosclerosis or reocclusion after a percutaneous intervention) and 4. secondary unstable angina – in condition with high oxygen demand and low supply (tachycardia, anemia) superimposed on a compromised coronary bed due to atherosclerosis. In NSTEMI the thrombus is called “white” since it is platelet rich.
In STEMI, the most common cause of obstruction is total occlusive thrombus with total compromise of downstream blood flow. In rare cases STEMI can be caused by total occlusion of a coronary vessel by a piece of tumor or other embolic material (intracardiac thrombus), coronary spasm, cocaine abuse or congenital anomalies. In STEMI
the thrombus is called “red” thrombus and represents classical thrombus.
The approach to the STEMI therefore is somewhat more urgent since the chances of finding and treating total thrombotic occlusion in the coronary arteries is higher. However the chances of finding thrombus in the coronary arteries in the unstable angina patient and NSEMI are still high but the thrombus is not usually totally occlusive. Vigilance and focused and emergency attention to all the entities is prudent.
Principles
Chest pain is “an unpleasant sensory and emotional experience associated with actual or potential tissue damage and mediated by specific nerve fibers to the brain where its conscious appreciation may be modified by various factors.” In the case of the heart, insufficient supply of oxygen to the myocardium is the underlying principle, but the exact mechanisms of how the pain is caused are not well understood. We will focus on the underlying anatomical and pathophysiological principlesof blood flow.
At its most basic, the the coronary artery is a tube that transports fluid.

A Simple Tube |
| 72845.800 aorta flow principles structure laminar flow turbulent flow normal Davidoff Art Courtesy Ashley Davidoff MD 72835.800 72839 72831.800 72845.800 49483b01 |
The coronary artery is more complex than a simple tube for many reasons. Its course is not straight but is curved. Many branches of varying size, changing resistances, and branching pattern with varied angles of origin make the flow more complex. The muscular and elastic nature of its walls and the viscosity and complexity of blood also contribute to the complexity of its physiology, pathophysiology, and imaging.
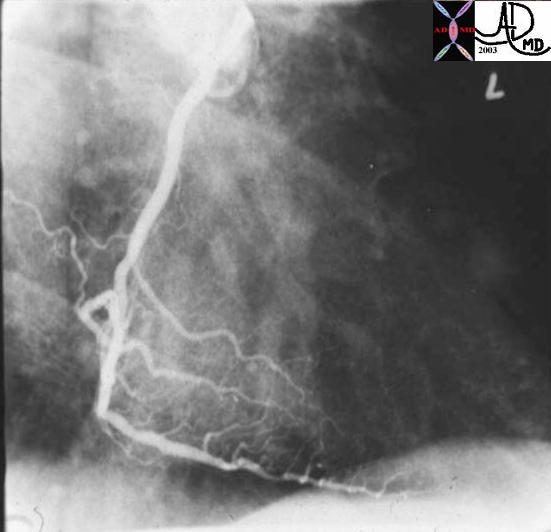
Angiogram of a Normal Right Coronary Artery |
| This is a normal right coronary angiogram in the right anterior oblique projection showing widely patent vessels. Note that that it is not a simple straight tube but a branching structure with curving branches that come off at a variety of angles, that are subject to pulsatile flow
Courtesy Ashley Davidoff MD. 07045 code heart artery coronary LAD circumflex obtuse marginal OM normal septal LM left main diagonal imaging radiology angiography |
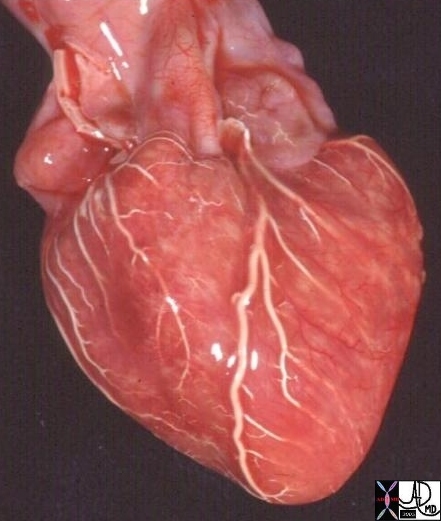
Normal Coronary Arteries – Post Mortem Specimen |
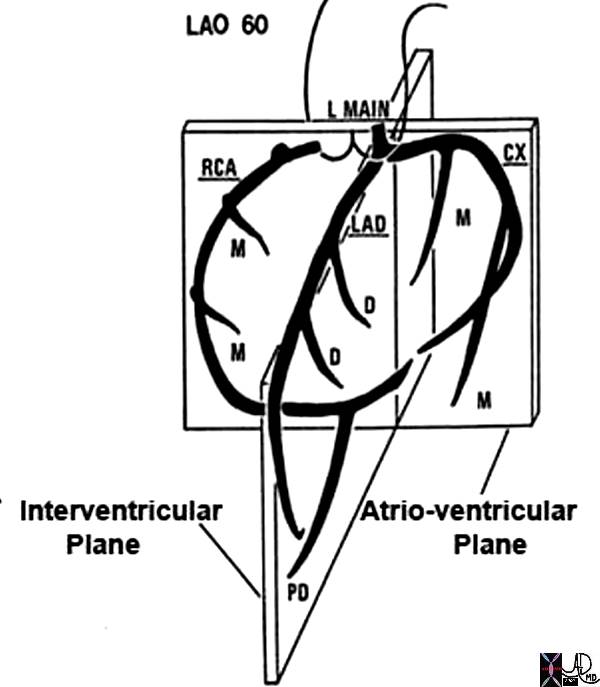
| The coronary arteries of the post mortem specimen have been injected with barium and thus look white. This anterior view shows the left anterior descending artery (LAD) coursing between the two ventricles to the apex of the heart. The diagonal branches (D) arise at acute angles and course over the left ventricle. The marginal branches (M) from the right coronary artery (RCA) are seen overlying the right ventricle, and the marginal branches (M) from the left coronary artery) are seen overlying the left ventricle.
The drawing on the right is provided to give a perspective of where these vessels lie.
15009b01 heart cardiac coronary artery LAD left anterior descending artery first diagonal artery second diagonal artery third diagonal artery fourth diagonal artery fifth diagonal artery anatomy normal post mortem angiogram angiography A-P projection Courtesy Ashley Davidoff MD
|
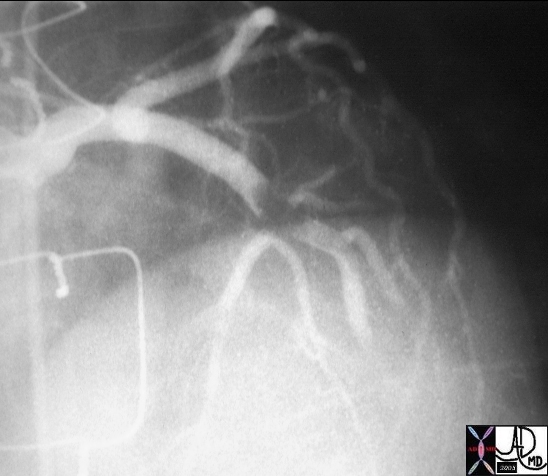
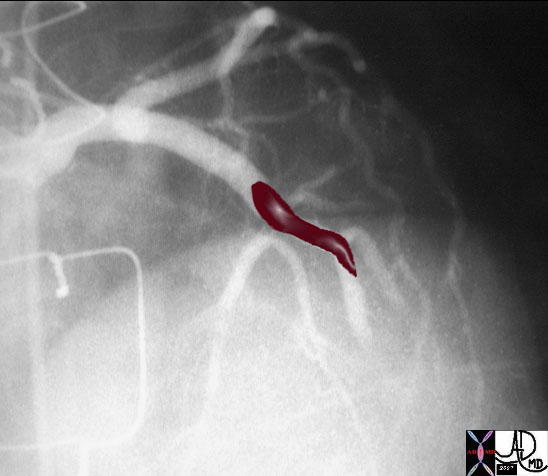
| Thrombus in LAD |
| The first image on the left shows a coned down RAO projection if the LAD in a patient with acute thrombosis of the left anterior descending artery. In the second image the thrombus in the artery is overlaid in maroon.
16950 16950b02 Courtesy Ashley Davidoff MD |
Applied Anatomy
Blood supply to the heart is provided by three coronary arteries: Right Coronary Artery (RCA), Left Anterior Descending (LAD) and Left Circumflex (LCx). The LAD and LCx usually originate from Aorta via a common trunk called Left Main (LM). If high grade Left Main stenosis is demonstrated on a catheterization patient may undergo bypass surgery as method of revascularization as opposed with percutaneous stenting. After a short course, left main divides into LAD and LCx. LAD is usually the biggest coronary artery and continues the traject of LM in the anterior interventricular groove down to the cardiac apex providing blood supply to anterior wall of the left ventricle. Septal branches (S1, S2, S3) originate form LAD and they provide blood supply for the anterior aspect of interventricular septum. Diagonals (Diagonal 1, Diagonal 2 and Diagonal 3) are also major branches of the LAD which supply the lateral wall of left ventricle. Left Circumflex takes off from Left Main at a sharp angle, and travels around the base of the heart towards the posterior aspect of the heart providing blood supply for the posterior wall of the heart. Major branches of Left Circumflex are Obtuse Marginals arteries(OM1, OM2, OM3). Right Circumflex Artery originates independently from Aorta and supplies the inferior wall of left ventricle. Acute marginal branches (A1, A2, A3) originates from proximal segment of RCA and supply the right ventricular wall which explain why an inferior wall MI (where the culprit vessel is RCA) can be associated with right ventricular infarction. In 60% of patients RCA also provides blood supply to sinus node and this fact explain why sinus bradycardia can be seen in patients with acute inferior MI (RCA infarct). The Posterior Descending Artery (PDA) is the biggest bifurcation of RCA running in the posterior interventricular groove and supplies the posterior aspect of the interventricular septum. Atrioventricular nodal branch originates from distal RCA after the PDA take-off and supply atrioventricular node explaining the presence of different AV node conduction abnormalities associated with inferior MI.
Applied Physiology and Pathophysiology
Pathophysiology of Acute Coronary Syndrome
Acute coronary syndrome represents the progression of stable coronary artery disease to unstable disease. At tissue level, atherosclerosis of the coronary arteries is the predisposing factor to an acute coronary event. Fissure or rupture of vulnerable atherosclerotic plaque is the central event leading to an acute coronary syndrome. Plaque rupture can be triggered by physical exertion, increase in cardiac contractility, tachycardia, high blood pressure and possibly vasoconstriction. Rupture of the plaque leads to platelets activation and aggregation and activation of clotting cascade, resulting in formation of a thrombus. If the thrombus causes total occlusion of the coronary artery, then ST segment elevation MI occurs. If the thrombus causes only incomplete obstruction of the coronary artery, then unstable angina or NSTEMI occur.²

The Normal Arterial Wall |
| This image shows normal circulating lipoproteins in the circulation. The intima is intact and the wall is normal. The yellow globules represent lipoprotein molecules in the lumen. The thin untainted intima or endothelium is seen on the luminal surface, and the normal media (chocolate brown) layer is seen superficial tot hw adventitia (golden yellow layer).
Courtesy Ashley Davidoff MD. 33789 Davidoff art |
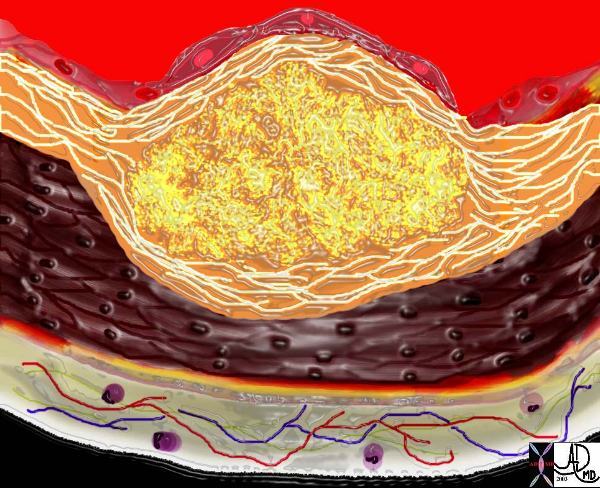
| An Atherosclerotic Plaque |
| The diagram shows the atherosclerotic lesion in the subepithelial layer of the intima which is bulging both toward the media and toward the lumen. There is a central core of fat and necrotic debris, surrounded by fibrous elements which give the plaque its hardness to the feel. The accumulation of fibrous tissue heralds an advanced atherosclerotic lesion. 33801b Courtesy Ashley Davidoff MD. code heart artery atherosclerosis Davidoff art |
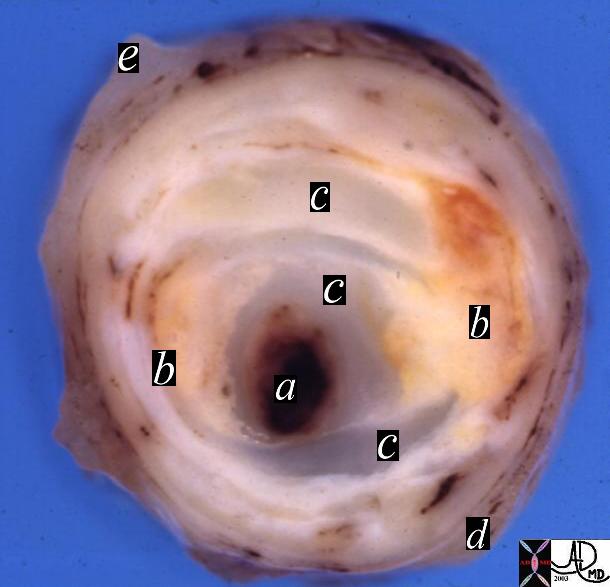
Stenosis of the Coronary Artery
|
| 13410b heart cardiac artery + coronary artery fibrocalcific plaque calcification narrowing stenosis dx atherosclerosis + grosspathology Courtesy Henri Cuenoud MD a= lumen or thrombus dx atherosclerosis + grosspathology Courtesy Henri Cuenoud MD b= cholesterol plaque c= fibrous cap d= media e= adventitia |
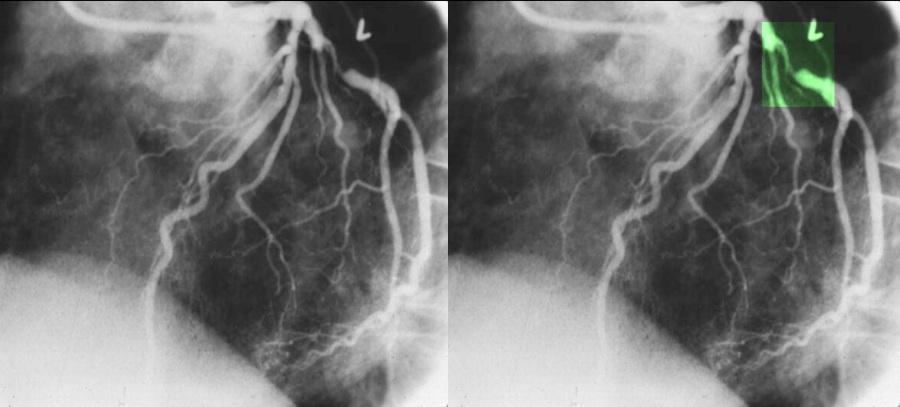
Atherosclerosis of the Right Coronary Artery |
| Coronary artery disease is the most common disorder that effects the heart mostly as a result of atherosclerosis which usually causes narrowing or obstruction of the vessel, but sometimes causes dilatation. In this instance both narrowing and dilatation is noted. The injection of the right coronary artery is in RAO view with two narrowings outlined in green. The first narrowing is of moderate degree, and the second is mild. A narrowing of 70% is considered hemodynamically significant. Courtesy of Ashley Davidoff M.D. 32140 |
Atherosclerotic plaque is seen radiologically in two forms. Fibrous plaque as seen in the angiogram above is characterized as a focal narrowing of the vessel while calcified plaque is better characterized by CT scanning and is exemplified below on the CT angiogram below. The fat containing plaque is the most dangerous plaque but it is not routinely identified on current imaging techniques.
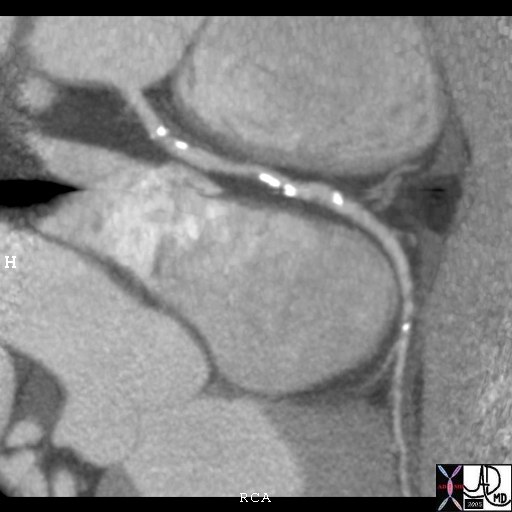
Calcified Plaques in the Right Coronary Artery |
| 44247 heart cardiac artery coronary artery heart cardiac artery coronary artery RCA fx calcification dx mild atherosclerosis imaging radiology CTscan Courtesy Ashley Davidoff MD Jeffrey Mendel MD |
Development of athersoclerosis reduces the lumen of the coronary vessel and limits the appropriate increase of myocardial perfusion during situations with high oxygen demand as exercise/high emotional stress or even at rest if high degree of stenosis.When the stenosis of the vessel is equal or greater than 75%, then anginal symptoms occur during those situations of high demand. When the vessel stenosis is greater than 80%, anginal symptoms may occur at rest. The degree of stenosis has a higher clinical relevance in pathophysiology of chronic angina. On the other hand, an acute coronary syndrome can result from an acute rupture of a plaque that doesn’t produce a flow limiting stenosis.Such lesions may not qualify as “significant” by arteriography based on the degree of stenosis only.
In the left main coronary artery a 50% lesion is considered hemodynamically significant. Clinical decisions regarding treatment of arterial lesions are based on the degree of stenosis.
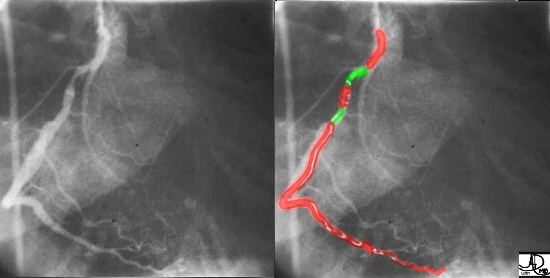
>70% Stenosis of the Left Circumflex Coronary Artery
|
| The coronary angiogram in the LAO projection shows a severe proximal stenosis (>70%) of the circumflex with some post stenotic dilatation. The second image has a green overlay indicating the region of disease with the post stenotic dilatation.
Courtesy Ashley Davidoff MD. 07023c |
Acute coronary syndrome represents the progression of stable coronary artery disease to unstable disease. At tissue level, atherosclerosis of the coronary arteries is the predisposing factor to an acute coronary event. Fissure or rupture of vulnerable atherosclerotic plaque is the central event leading to an acute coronary syndrome. Plaque rupture can be triggered by physical exertion, increase in cardiac contractility, tachycardia, high blood pressure and possibly vasoconstriction. Rupture of the plaque leads to platelets activation and aggregation and activation of clotting cascade, resulting in formation of a thrombus. If the thrombus causes total occlusion of the coronary artery, then ST segment elevation MI occurs. If the thrombus causes only incomplete obstruction of the coronary artery, then unstable angina or NSTEMI occur.²
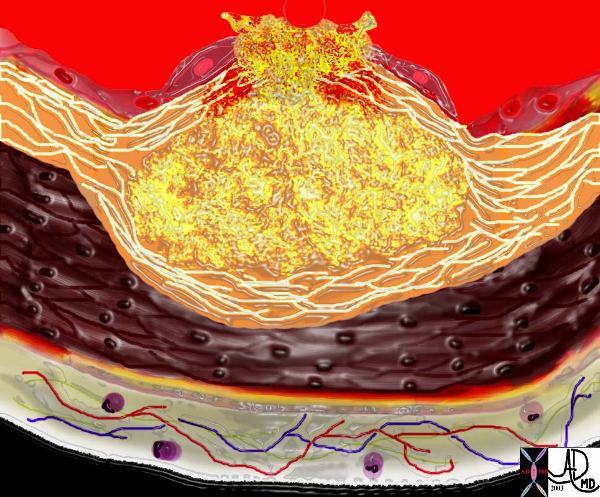
A Ruptured Atherosclerotic Plaque |
| This diagram shows denudation of the endothelial layer with exposure and rupture of the contents of the atherosclerotic plaque in volcanic fashion into the lumen. This event is catastrophic and can result in acute thrombosis and may even be a fatal event.
33801d Courtesy Ashley Davidoff MD. code heart artery atherosclerosis atheroma vulnerable plaque drawing Davidoff art |
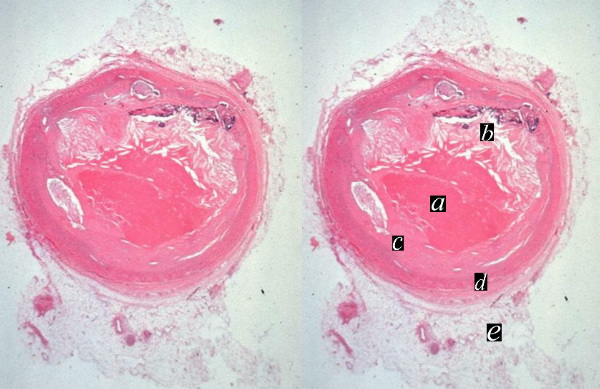
Acute Thrombus on Atherosclerotic Plaque |
| The histopathological specimen shows acute thrombosis of the coronary artery with red homogeneous thrombus (a) filling the lumen. “b” overlies the cholesterol plaque while c is fibrous plaque. D is the muscularis and e is the adventitia.
13318c heart cardiac coronary artery fx narrowing fx stenosis fx plaque fx eccentric plaque a= thrombus b= cholesterol plaque c= fibrous plaque d= media e = adventitia dx atherosclerosis thrombosis histopathology Courtesy Dr Isabelle Joris |
Causes and predisposing factors
Nonmodifiable risk factors for cardiovascular disease are increasing age, male sex, ethnicity and heredity (family history). Women’s risk of cardiovascular disease increases after menopause. On average women manifest the disease 10 years later then the men. However, at age 70, the risk of cardiovascular disease is equal in men and women. In women, concurrent diabetes confers a higher risk compared with men with diabetes.³
Family history plays an important role in the risk of developing CVD later in life. Having a male relative who experienced MI before age 55 or a female relative with MI before age 65 places a person at a higher risk of cardiovascular disease.4
Also, different ethnic groups have different risks, with Mexican Americans, American Indians and Hawaiians being at higher risk for CVD. The incidence of hypertension is higher on African Americans compared to Caucasians as well as their risk for CVD.
Modifiable risk factors are diabetes, HTN, chronic kidney disease, smoking, dyslipidemia, physical inactivity, and obesity. Lifestyle changes and drug therapies can modify risk factors leading to decrease morbidity and mortality in such patients.
Clinical Manifestation of Acute Coronary Syndomes
The patient’s history remains of critical value in diagnosis of an acute coronary syndrome despite advances in biochemical markers, EKG and imaging modalities.
The classic symptom of an acute coronary syndrome is chest pain. Typically, pain is retrosternal or precordial and may radiate to the neck, jaw, shoulders, left arm or back (interscapular area). Pain is due to irritation of nerves endings in ischemic or damaged myocardium, but not necrotic yet. Pain resembles to the chronic stable angina pain however in most patients is more severe in intensity, lasting for more than 30 minutes and more frequently for a numbers of hours, usually not relieved by nitroglycerin or by rest. Pain is being described as pressure, heaviness, tightness, squeezing or crushing and is triggered by physical activity, emotional stress or circadian rhythm. Usually pain responds to opiates, morphine in particular. Symptoms associated with the pain can be nausea, vomiting, palpitations, diaphoresis and subjective shortness of breath. Nausea and vomiting are more frequent with inferior location of myocardial infarction. When the pain is located in the epigastric area and is associated with nausea and vomiting, the clinical picture can be confounded with acute cholecystitis or peptic ulcer. Once again, the characteristics of the chest pain with myocardial infarction is very similar to that of unstable angina, but has been described as more severe in most cases.
Many patients experience less typical symptoms with myocardial infarction as a feeling of profound weakness and fatigue, dizziness, palpitations, cold perspiration without associated chest pain. Also, pain occurring in other location then the chest, i.e. epigastric, numbness sensation of the chest or left arm, tingling sensation in the left wrist, hand or fingers, stabbing or burning pain, lightheadedness, or “indigestion” symptoms have been reported as initial presentation of myocardial infarction. These “atypical” symptoms of MI occur more frequently in women, elderly and in patients with diabetes mellitus, hypertension, heart failure and stroke.5, 6
In elderly, frank syncope, as a sign of left ventricular failure secondary to massive myocardial infarction can be the initial presentation of STEMI.
Multiple studies have reported lack of typical chest pain in as many as 33% of patients experiencing an acute coronary syndrome.7
Differential diagnosis of ACS
The chest pain as a result of an acute coronary syndrome should be differentiated by other causes of chest pain. Sometime the pain from MI may simulate the pain of acute pericarditis therefore the big distinction is between the ischemic pain and pericardial or pleuritic chest pain as seen with acute pericarditis or pulmonary pathology respectively (i.e., pulmonary infarct, pulmonary emboli). Major clinical difference is that respiratory movements, taking a deep inhalation or coughing, aggravate both pleuritic and pericardial chest pain and the pain is reported as sharp, knife-like, which distinguishes from deep, steady, dull pain of MI.
Aortic dissection also causes chest pain which is described by the patients as “ripping” or “tearing” sensation radiating to the back and has maximal intensity shortly after onset. On physical examination, aortic dissection can cause absence of one of the major arterial pulse and blood pressure discrepancy between both arms.
Costochondritis can also mimic ischemic chest pain but the former pain is sharp and associated with marked localized tenderness.
Esophageal spasm also should be in the differential on MI.
Physical exam
Patients with MI appear in distress, restless. They can have skin pallor and cold perspiration. Also patients experience difficulty breathing with severe left ventricular failure.If cardiogenic shock is present, the skin is cool and clammy with mottled appearance over the extremities and patients may present with confusion and disorientation secondary to cerebral hypoperfusion.
Vitals signs, including BP in both arms, pulse and temperature should be checked. Most patients with STEMI develop fever in response to tissue necrosis in the first 24-48 hours after the onset of infarction.
The heart rate can vary from bradycardia – typically in inferior STEMI to sinus regular tachycardia or irregular tachycardia with frequent premature ventricular beats. Blood pressure is normal in majority of uncomplicated MI, however in previously normotensive patients in the first initial hours of the event the blood pressure may be elevated due to high adrenergic state. In patients with massive STEMI and cardiogenic shock, the arterial blood pressure falls acutely to level below 90 mm Hg. Hypotension and bradycardia can occur also secondary to increased vagal tone in inferior wall MI in the absence of left ventricular failure or cardiogenic shock.
Cardiac examination of patients with MI may be unremarkable or there may be clinical signs suggestive of ischemia or heart failure as a result of the acute ischemic event. On auscultation, the heart sounds may be decreased in intensity acutely. New systolic murmur can be heard as a result of transient/persistent mitral regurgitation caused by acute ischemic papillary muscle dysfunction or rupture. Paradoxical split second heart is present in extensive myocardial dysfunction and subsequent left bundle branch block (LBBB). Also, third cardiac sound can be present, jugular vein distension or rales on lung auscultation if severe heart failure develops secondary to MI. Peripheral vessels should be examined for bruit or pulse deficit.
A careful physical examination may help to rule out aortic dissection, pneumotorax, pericarditis, cardiac tamponade or congestive heart failure.
Labs
The diagnosis of ACS/MI requires at least two of the following three criteria according with World Health Organization: history consistent with ischemic type of chest pain, changes on serial EKG tracing and rise and fall in cardiac markers.8
EKG
EKG is of crucial importance in the diagnosis of an MI. The EKG changes can help with the distinction between STEMI and NSTEMI however is less useful to differentiate NSTEMI from UA. In UA the EKG can show ST segment depression in the leads corresponding the myocardial territory supplied by a coronary artery. In NSTEMI, there is typically ST depression, T wave inversion or nonspecific ST-T changes.
In STEMI, the first sign of an acute event could be acute elevation of T wave which is called “hyper acute T wave”, followed by ST segment elevation, then normalization of ST segment with development of T wave inversion and Q wave. When an ACS is being suspected by the history the EKG should be repeated every couple of hours, as these changes are dynamical. Also, the EKG should be compared with an old one if available.
The changes of STEMI can be masked by the existence of LBBB. Any presumed new LBBB on EKG with a clinical presentation suggestive of MI should be managed as an STEMI.
EKG changes can be used to predict the location of MI.
Location Leads Vessels
Anterior V2-V4 LAD
Anteroseptal V1-V4 LAD
anterolateral V1-V6, I, aVL LAD, Diagonal
inferior II, III, aVF RCA, Circumflex
lateral I, aVL, V5-V6 Circumflex, Diagonal
Posterior tall R in V1-V3 RCA
Right ventricle right V4 RCA
RCA= right coronary artery
LAD= left anterior descending
Blood Tests– Cardiac Markers
The creatine kinase (CK), the creatine kinase MB (CK- MB) and troponins (I or T) are currently used as biochemical markers of cardiac damage. The cardiac enzymes should be checked on presentation and every 8 hours until peak. The troponins are the gold standard for diagnostis of MI as they have higher sensitivity and specificity. Troponins can be detected in blood 4-6 hours post injury, peaks at 24 hours and they remain elevated in serum for 7-10 days.
CK – MB is less sensitive and specific as it can be released from skeletal muscle as well, tongue, diaphragm, intestine, uterus, and prostate. Both total CK and CK-MB are detectable in blood at 4-6 hours, peak at 24 hours and returns to normal in 2-3 days.
Myoglobin is less used in diagnostic on MI but can be used when an urgent diagnostic is needed in the absence of EKG changes. Myoglobin appears in the serum 30-60 minutes after the onset of symptoms but falls quickly. It is less specific than troponin but has higher sensitivity, especially when used in conjunction with troponins.
Keep in mind that cardiac enzymes can be positive in a patient with chest pain caused by a massive pulmonary embolism which causes strain on the right ventricle releasing cardiac enzymes from right ventricular ischemia or infarct. Also, acute aortic dissection which involves the origin or coronary arteries can cause elevation of cardiac enzymes. Myopericarditis also is associated with elevation of cardiac enzymes. Usually the character of chest pain/EKG can help with the distinction between ischemia as a cause of elevation of cardiac enzymes and other pathologies (see Differential diagnostic section), however there are multiple situations when the clinical history is vague or unavailable so the diagnostic of MI is challenging and implies using different imaging modalities which are not routinely used (see Imaging section).
Cardiac enzymes should be checked on every patient with history suggestive of acute coronary syndrome however in patients with ischemic chest pain and ST elevation segment on EKG or new left bundle branch block the decision for reperfusion therapy should not be delayed waiting on cardiac enzymes result as they may remain negative for the first few hours. Cardiac enzymes are more important in risk stratification and decision making in patients with NSTEMI and actually help making the distinction between NSTEMI and UA.
Other blood tests
Lipid profile (serum total cholesterol, LDL cholesterol and HDL cholesterol) should be obtained at the time of initial presentation as the level may be falsely low after 24 hours since presentation. For patients admitted 24- 48 hours after the acute event, a lipid profile should be done 8 weeks after infarction has occurred.
Hematological findings
Elevation of white blood cell count develops 2-4 hours after the onset of chest pain and reaches a peak 2-4 days after infarction and returns to normal in 1 week The usual peak ranges from 12.000- 15.000/ml but can be as high as 20.000 is patients with large STEMI.
Markers of inflammation like erythrocyte sedimentation rate (ESR) and C reactive protein (CRP) can be also elevated after the first few days after infarction and they may remain elevated for few weeks.
How quickly are these studies done? When do results come back of blood testst Do you wait for these results before deciding on cath ? ie how long does it take for enzymes to come back?
Imaging
Usually the diagnosis of acute coronary syndrome can be made easily if the clinical history, physical examination, EKG and serum markers (ie cardiac enzymes) are all suggestive of ischemia. Keep in mind that cardiac enzymes can be detected in the blood only 4-6 hours after the ischemic event so if the patient presents to ER immediately after the chest pain episode, the first set of cardiac enzyme may be negative even if the patient is experiencing a true MI. Further diagnostic imaging tests to rule out other possible causes of chest pain are usually done during evaluation of a patient with a possible acute coronary syndrome when the history, physical exam and EKG tracing are not totally consistent with an ischemic event. If the first set
Preliminary Studies
Portable CXR should be done routinely during evaluation of a patient with chest pain. It may help rule out some other causes of chest pain than ischemia: aortic dissection which would show up on CXR as a widened mediastinum, pneumotorax, ribs fracture. Also, CXR in patients with cardiogenic shock as a result of an acute MI will show acute pulmonary edema with proeminent pulmonary vascular markings and diffuse alveolar infiltrates. However the radiographic appearance of pulmonary edema need up to 12 hours of elevated left ventricular end diastolic pressure to develop and the pulmonary changes may persist on CXR for 2 days after the ventricular filling pressures have normalized. The presence of congestion on CXR has been shown to confer a worse prognosis in both STEMI and STEMI.

Normal (left) and Pulmonary Edema (right) |
| The patient with acute myocardial infarction may have a normal chest examination but if there is extensive myocardial damage to impede pump function, pulmonary edema (right) results.
46813c01.800 chest lung pulmonary edema alveolar edema batwing distribution drowned lung dax cardiogenic shock normal CXR plain film of the chest Davidoff MD |
Non Invasive Imaging
Transthoracic echocardiogram can be useful in diagnosing MI especially in patients with chest pain suggestive of ischemia but without characteristic EKG changes. Regional wall motion abnormalities in a patient with chest pain make support the diagnostic of myocardial ischemia. Echocardiography is also useful to rule out aortic dissection by identifying an intimal flap. Transesophageal echocardiogram may be superior to standard echo in identifying aortic dissection. Also transthoracic echo can be used to evaluate right ventricular strain and dysfunction caused by massive pulmonary emboli. Color Doppler echocardiography is useful to identify the severity of mitral or tricuspid regurgitation secondary to MI and some potential mechanical complications of STEMI such as acute ventricular septal rupture, free wall rupture and cardiac tamponade. These complications usually occur 4-5 days after the acute MI.
Ruling out aortic dissection in the setting of an acute MI is of crucial importance as this is a major contraindication to fibrinolytic therapy. As mentioned above it can cause chest pain with cardiac enzymes elevation and ST segment elevation due to dissection involving coronary arteries. Surgery will be the only treatment option in that particular circumstance.
| Perfusion Echo Normal LV (left) and Abnormal LV (right) |
| This real time perfusion imaging echo of the heart(left image) shows the left ventricle of a 4 chamber view, and demonstrates homogeneous perfusion and normal conical shape of the LV. The image on the right shows a dilated left ventricle revealing a heterogeneous and abnormal perfusion of the myocardium.
Courtesy Philips Medical Systems 33117 33115 code cardiac heart imaging cardiac echo |
| Mitral Regurgitation |
| The 4 chamber view of the heart using pulse Doppler, as well as color flow Doppler shows color changes in the left atrium which is abnormal and is pathognomonic of mitral regurgitation. If this finding is a new finding in a patient with AMI it indicates severe structural damage. Papillary muscle rupture is one of the considerations in the setting of acute MI but it usually occurs about 8 days after the MI when the muscle is at its weakest.
Courtesy Philips Medical Systems 33119 cardiac heart echo mitral valve regurgitation MV MR imaging cardiac echo |
CT angiography is not part of routine diagnostic workup of MI. It can be used to rule out pulmonary embolism showing filling defect in the pulmonary vessels or dissection of aorta in patients with chest pain and EKG changes non diagnostic of MI.
Complications of STEMI
Cardiac Catheterization
Cardiac catheterization should be performed within 90 minutes from ER arrival in patients with ischemic chest pain and STEMI or new LBBB on EKG. Also patients with NSTEMI and high risk based on TMI risk score benefit of early invasive strategy with angioplasty versus conservative medical management. If cardiac catheterization is not available at the medical facility, patients with STEMI should be treated with intravenous thrombolysis within 30 minutes of arrival to the ER.
  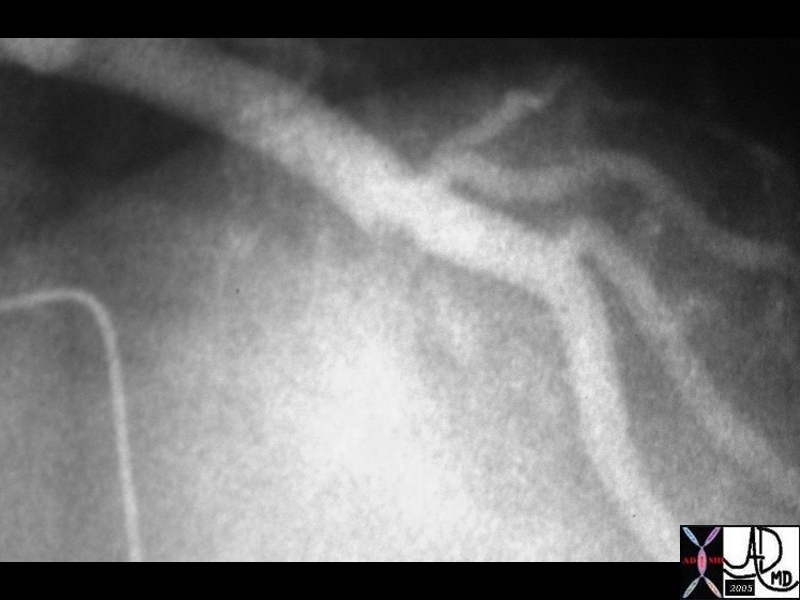 Thrombus in LAD then Thrombolysed Thrombus in LAD then Thrombolysed |
| The first image on the left shows a cned down LAO projection if the LAD in a patient with acute thrombosis of the left anterior descending artery. In the second image the thrombus in the artery is overlaid in maroon. Following thrombolysis *third image minimal irregularity of the endothelium is seen. This may be residual thrombus or the st of a plaque rupture
16950 16950b02 16956 cardiac heart coronary artery LCA thrombus acute MI Courtesy Ashley Davidoff MD |
Complications of STEMI
After an STEMI, patients can have mechanical complications (cardiogenic shock, free ventricular wall rupture, ventricular septal defect, papillary muscle rupture), arrhythmia (atrial fibrillation, ventricular tachycardia, ventricular fibrillation, sinus bradycardia, heart block), left ventricular aneurysm, left ventricular thrombus, and pericarditis. Mechanical complications usually occur 3-4 days after the acute event, they manifest as new onset hemodynamical instability (hypotension) and new murmurs on auscultation. Surgical repair is usually required.
Atrial fibrillation post MI occurs in 10-15 % of patients. It may require cardioversion if rapid ventricular response and hypotension occur. Otherwise, the ventricular rate should be controlled with beta blocker, calcium channel blocker or amiodarone.
Monomorphic ventricular tachycardia (VT) and ventricular fibrillation (VF) within 48 hours post MI does not carry bad prognosis. Antiarrhythmic (lidocaine, amiodarone) are used for monomorphic VT without hemodynamic compromise and cardioversion/defibrillation if ventricular fibrillation or pulseless VT. Electrolytes (potassium, magnesium) should be repleted (potassium >4 mEq/L, magnesium >2 mEq/L) and recurrent ischemia has to be rule out. If VT/VF persists for more than 3 days after MI and there is no evidence of recurrent ischemia, then an implanted cardio defibrillator is advisable.
Sinus bradycardia and different blocks can occur post MI and therapy can vary from atropine to temporary or permanent pacing according to duration of block and presence of symptoms (hypotension, mental status change due to brain hypoperfusion, decrease urine output due to kidney hypoperfusion, etc).
Left ventricular thrombus can occur in 20-40 % of patients with large antero-apical MI and requires anticoagulation with coumadin for 3-6 months.
Ventricular aneurysm and pseudoaneurysm can also complicate an STEMI and sometime require surgery. They can be easily identified on echocardiogram. Presence of persistent ST segment elevation on 12 leads EKG was classically thought to represent aneurysm formation but actually indicates a large infarct with regional wall motion abnormalities.
Pericarditis can occur after an MI and a rub can be heard on physical exam. Treatment consists of nonsteroidal anti-inflammatory, high dose aspirin and at the same time to decrease the anticoagulation medication (i.e. heparin, integrillin), as there is higher potential for bleeding.
After anSTEMI, patients can have mechanical complications (cardiogenic shock, free ventricular wall rupture, ventricular septal defect, papillary muscle rupture), arrhythmia (atrial fibrillation, ventricular tachycardia, ventricular fibrillation, sinus bradycardia, heart block), left ventricular aneurysm, left ventricular thrombus, and pericarditis.
Management
There are diagnostic and therapeutic priorities when evaluating patients with possible acute coronary syndrome.
The correct identification of the diagnostic of MI is of crucial importance and sometime can be a major challenge. Furthermore, identification of patients with high risk features will dictate the therapeutic plan.
The ultimate therapeutic priority is restoring the coronary blood flow and minimizing the sequelae of ischemia. In addition to restoring the coronary blood flow, pain control and achieving hemodynamical stability is part of the immediate management in ER.
Triage begins with taking a good history and assessment of symptoms. Besides the classical symptoms of chest pain, suspicion also should be raised by “atypical” symptoms (see clinical manifestation section). Physical examination with recording of vitals, EKG tracing within 10 minutes from triage, CXR and draw of cardiac enzymes should be done. Patient should have continuous telemetry EKG monitoring of the cardiac rhythm, blood pressure and oxygen saturation monitoring by pulse oximetry. If the patient is hypotensive, hemodynamic resuscitation with intravenous fluid (normal saline as bolus) should be given. Also they may benefit of vasopressor medication. If supraventicular tachycardia is present, metoprolol 5 mg iv every 5 minutes can be administered until heart rate is controlled provided the patient is not hypotensive. If ventricular tachycardia is present and patient is hemodynamically stable, antiarrhythmic therapy with lidocaine, amiodarone is appropriate. If unstable ventricular tachycardia or ventricular fibrillation, electrical defibrillation or cardioversion is advised.
Risk factors should be also taken into account when evaluating a possible acute coronary syndrome as they help with prognostic and with the decision where to admit the patient.
TIMI risk score is a useful clinical tool to stratify patient’s risk of adverse outcome following of diagnostic of NSTEMI/UA. The 7 risk factors are: age>65, more then 3 risk factors for CAD (see risk factors section), known past medical history of CAD, aspirin use, severe angina on presentation, ST segment deviation, and positive cardiac markers. Each of these 7 factors get 1 point, with a maximal score of 7. The higher TIMI risk on presentation, the higher risk of adverse outcome of NSTEMI/UA and more benefit from an aggressive therapeutic strategy. 9
The chance of finding a thrombus is not the element that influences therapy but rather the extent of myocardial damage secondary the obstruction, and the percentage of myocardial mass that can be salvaged by an early intervention. The entire clinical picture (patient stability, risk factors as outlined above) are important considerations when management decisions are being made. In STEMI, irreversible necrosis of the myocardium will occur if blood flow is not restored in a timely fashion as the blood vessel is totally occluded by the thrmobus. In NSTEMI the decision to go to cath lab relies heavily on the TIMI score.
Where the patient gets admitted to?
Patients with any of the following high risk criteria should be admitted to coronary care unit or step down unit:
age older than 75
ongoing chest pain at rest for more than 20 minutes
accelerating symptoms within last 48 hours
signs of pump failure (hypotension, lung rales, pulmonary edema) associated with ST segment depression or elevation on EKG
positive enzymes
Patient at intermediate risk with any of the following criteria should be admitted to either telemetry floor or step down cardiac unit:
prior history of coronary artery disease (prior MI, prior CABG, prior aspirin use)
history of cerebrovascular disease/peripheral vascular disease, age more than 60, without ongoing chest pain
T wave inversion on EKG and only slightly elevated troponin
Management of UA/NSTEMI
The management of NSTEMI/UA consists of pain control/adjunctive therapy and restoring the coronary blood flow (antithrombotic therapy +/- angiography). Based on the TIMI risk score (see below) a conservative strategy versus an invasive strategy may be chosen. Patients with TIMI risk score higher than 3 benefit from an early invasive strategy with percutaneous angiography.
If an invasive strategy is considered and angiography +/- angioplasty is being planned based on higher TIMI risk score (>3) on initial evaluation, then the patient may be started on Glycoprotein IIb/IIIa inhibitors drip (i.e. Integrillin) in addition to aspirin and heparin before the angiography. Clopidogrel (i.e. Plavix) can be deferred in the invasive strategy for increase bleeding risk.
Conservative management (anti-thrombotic therapy) is usually chosen for low risk patients (TIMI risk score 0-2) and consist of consist of aspirin (162-325 mg orally- first dose should be crushed/chewed to allow faster absorption form oral mucosa), clopidogrel with 300 mg first loading dose then 75 mg daily. Patient should be started on unfractioned heparin drip or low molecular weight heparin. Patient will have a noninvasive stress test before discharge home. If the stress test is positive for ischemia, then patient should undergo an angiography. If patient develops recurrent chest pain during the hospital stay then angiography will be reconsidered.
Pain control can be achieved with placing the patient on oxygen regardless the level of oxygen saturation on pulse oxymetry, nitrates administered sublingual, oral, topical or intravenously. Adjunctive therapy consists of beta blocker (metoprolol 5 mg iv every 5 minutes provided heart rate>60, blood pressure >100, no heart block or severe brochospasm). Calcium channel blockers (i.e. Verapamil, Diltiazem) can be used to slow down the heart rate and for high blood pressure if patient cannot tolerate beta blocker due to coexistent morbidities (severe asthma/COPD). Morphine is also used to relieve chest pain not responding to nitrates and also will help with relieving the signs of pulmonary congestion.
It is very important to monitor for symptoms and signs of bleeding while the patient is on the above anti-thrombotic medications, like external bleed which would be obvious versus internal bleed which would manifest as hypotension, tachycardia, drop in hemoglobin and hematocrit, etc.
Management of STEMI
If the EKG changes are consistent with STEMI in the context of clinical history consistent with an acute coronary syndrome, then the reperfusion therapy is the core piece of management. Time is crucial in the management of STEMI and the outcome is better as the time to achieve successful reperfusion is shorter.
There are 2 methods to achieve restoration of the blood flow in STEMI depending on the level of the medical facility: fibrinolysis and percutaneous coronary intervention (PCI).
Fibrinolysis therapy is chosen when there is no catheterization lab available in the facility and the time to transfer the patient to another facility may take to long. Fibrinolytic therapy should be given within the first 30 minutes since arrival at the hospital (door to needle time<30 minutes) if the patient had symptoms of an acute MI for less than 12 hours with new LBBB or STEMI on EKG. The benefit of fibrinolytic therapy is less clear beyond the first 12 hours since the onset of symptoms.
There are absolute contraindications to fibrinolytic therapy (as any prior intracranial bleeding, ischemic stroke or head trauma within 3-6 months, active bleeding, intracranial neoplasm, suspected aortic dissection) and relative contraindications (systolic blood pressure> 180, INR>2, recent internal bleed within 2-4 weeks, prolonged CPR> 10 minutes, trauma or major surgery in 2-4 weeks, pregnancy etc). The trombolytic agents are Tenecteplase (TNK), Alteplase (TPA), and Streptokinase (SK). In addition to fibrinolytic therapy, patients should receive 162- 325 mg of aspirin (chewed or crushed) and heparin (low molecular weight heparin being better then unfractioned heparin in patients with age <75 and creatinine<2).
Primary percutaneous coronary intervention (PCI) should be done is 90 minutes since arrival to ER (door to balloon time = 90 minutes). Primary PCI is superior to fibrinolysis however there are many hospitals without a cath lab available 24/7. PCI should be considered especially if cardiogenic shock, anterior MI or heart failure secondary to STEMI. Patients in which primary PCI will be done should also be started on antithrombotic therapy, i.e. aspirin, unfractioned heparin drip and glycoprotein IIb/IIIa inhibitors drip.
In addition to fibrinolytic therapy, for the pain control in STEMI please refer to the management of NSTEMI/UA (i.e., oxygen, nitrate, morphine, beta blocker).
TIMI Risk Score
TIMI risk score is a useful clinical tool to stratify patient’s risk of adverse outcome following of diagnostic of NSTEMI/UA. The 7 risk factors are: age>65, more then 3 risk factors for CAD (see risk factors section), known past medical history of CAD, aspirin use, severe angina on presentation, ST segment deviation, and positive cardiac markers. Each of these 7 factors get 1 point, with a maximal score of 7. The higher TIMI risk on presentation, the higher risk of adverse outcome of NSTEMI/UA and more benefit from an aggressive therapeutic strategy. 9
Similarly, there is a TIMI risk score for STEMI taking into account age, history of DM/HTN/angina, blood pressure< 100 mm Hg, HR> 100, heart failure on clinical exam (i.e. lung rales), weight <67 kg, anterior location of MI or new left bundle branch block and delay to therapy of more then 4 hours. Again, the higher the score, the higher the mortality would be from the acute STEMI.10
Red Flags
As mentioned above the diagnostic of an acute myocardial infarction can be sometime very challenging even for the health care professionals so the patient’s awareness of red flags for an acute cardiac ischemic leading to a emergency room visit can be potentially life saving. Presence of any of the following conditions/characteristics represents a red flag which should raise the patient or the medical staff suspicion for an MI.
- Personal risk factors (HTN, DM, hyperlipidemia, smoking etc)
- First episode of chest pain
- Chest pain at rest occurring in patient with history of exertional chest pain
- Chest pain not relieved by nitroglycerin
- Chest pain lasting longer than 20 minutes
- SOB, “indigestion symptoms” like nausea or vomiting, diaphoresis, left arm numbness (see clinical manifestations),
- EKG changes (ST elevation or depression)
- Positive cardiac markers of ischemia – those can be negative on first blood draw!!!
Patient information
Experiencing an MI can be a very stressful event in patient’s life so during the initial ER presentation providing appropriate information about the diagnostic, therapeutic options, expected complications from both the acute event itself and from therapy is of crucial importance.
The initial focus of the patient presenting with a suspected MI is one of confirming the diagnosis using a combination of blood tests, EKG patterns, and possibly an angiogram. This is sometimes obvious immediately, and sometimes takes a few hours.
Once the diagnosis of STEMI is made, patient and family should be aware of the two main therapeutic options available; intravenous fibrinolytic therapy or cardiac catheterization with local thrombolysis, angioplasty and vascular stenting (based on individual hospital level). Patients should be informed of major complications of each option, i.e. bleeding with fibrinolysis and groin hematoma, bleeding, and renal failure with cardiac catheterization. They should be informed that prognosis is dependent on the time to reperfusion; the sooner the better.
Patient and family should be aware that the first 48 hours after a massive heart attack can be critical, with most arrhythmic complications occurring during this period. Also, they should be informed that fatal mechanical complications can occur with ST segment elevation MI even few days later after the acute event. The extent of the cardiac muscle damage will dictate the overall prognosis and this information can be obtained with an echocardiogram of the heart to assess the residual cardiac function after the MI.
Later on during the hospital course the caregiver should inform the patient about the overall prognosis and impact on lifestyle. Secondary prevention strategy aims to help decrease the chance of a future event.
The time before discharge is an important opportunity to communicate risk factor modification. Patients should be informed about the importance of achieving optimal weight, daily exercise, appropriate diet, smoking cessation, blood pressure control, lipid control and hyperglycemia control for diabetic patients. The importance of medication compliance must be reinforced. Long term mortality improvement has been shown with antiplatelet therapy, aspirin, clopidogrel for prevention of thrombosis, beta blockers for hypertension and angina, statins for hyperlipidemia, and angiotensin converting enzyme inhibitors for heart failure and hypertension.
Before discharge from hospital after an STEMI every patient should receive appropriate counseling regarding the level of physical activity they can safely perform. Initial physical activity should consist of ambulation with multiple daily rest periods. Patients should avoid isometric exercise such as lifting and any activities that evoke symptoms. The activity level should be gradually resumed as convalescence progresses. Regarding resuming sexual activity, this should be done after successful completion of a modified stress test. Patients should receive prescriptions for nitroglycerine and be instructed as to how and when to use the medication and also detailed information on all prescription medications they should take. Patients should be also be aware of all atypical symptoms of MI and have a low threshold to seek medical attention in the future. Cardiac rehabilitation programs for patients with STEMI are also available and gradual increase in the level of physical activity under direct supervision of qualified personal is recommended.
Given the association between depression and STEMI, psychological counseling should be offered to these patients.
Routine office visits every 4-6 months are suggested for the first year after the index event and annual visits afterwards. With every visit, the physician should address 5 questions:
- Has the patient decreased the level of physical activity since the last visit?
- Did the patient experienced any chest pain since last visit and if yes, in which circumstance and what are the characteristic (duration, severity, level of activity to provoke it)?
- Is the patient compliant and tolerating therapy?
- Has the patient attempted to decrease the risk factors for cardiac disease?
- Any new comorbidity that may increase the risk of ischemic heart disease?
Patient should be provided with web address where they can find additional information about heart disease:
Conclusion
“Chest pain suspect MI” is an emergency and not a straightforward recipe algorithm. The diagnosis is not always clear, the potential life threatening complications are real and anxiety provoking, and the treatment options are often time limited and based on quick and accurate diagnosis. All patients in this category need an urgent focus. To this end the first step is diagnosis, and although clinical presentation is highly suggestive, initial confirmation is based on EKG and blood tests. A first draw of the blood tests may be falsely negative and the EKG is also not always reliable. It is important to distinguish between STEMI and NSTEMI because the entities are approached differently.
The major difference pathologically between the two entities is that STEMI is usually caused by totally occlusive “red” thrombus while in the unstable angina group and NSTEMI the most common pathology is “white” platelet rich thrombus which is generally non occlusive.
The approach to the STEMI therefore is urgent catheterization to confirm the presence of occlusive thrombus and to institute thrombolysis. NSTEMI is also life threatening but is usually managed conservatively with medication, although angiography usually is performed during the admission. Although the STEMI has initial higher mortality due to a larger area of infarct and many potential complications, the one year survival is equal in both STEMI and NSTEMI. Patients with NSTEMI have a smaller size of infarct and decreased early mortality, but they are at higher risk for persistent angina, re-infarction and death within several months.
- National Library of Medicine
(www.nlm.nih.gov/medlineplus/healthtopics.html)
- National Heart, Lung, and Blood Institute
- American Heart Association
References
- Luepker RV, Apple Fs et al: Case definitions for acute coronary disease in epidemiology and clinical research studies. Circulation 108:2543, 2003
- Yerem Yeghiazarians et al, NEJM, Jan 13, 2000
- Legato MJ.Women’s health, Int J fertile. 1998; 43 (2): 65-72.
- American Heart Association. Heart and Stroke facts. Dallas, 2003
- National Heart Attack Alert Program Coordinating Committee Working Group. Educational strategies to prevent prehospital delay in patients at high risk for acute myocardial infarction. Bethesda , National Institutes of Health, National Heart, Lung and Blood Institute, 1997
- Penque Set al, Women and coronary disease. Am J Crit.Care, 1998; 7 (3): 175-182
- Canto JG, Shlipak MB et al. Prevalence, clinical characteristics and mortality among patients with myocardial infarction presenting without chest pain
- Pedoe-Tunstall H et al: Myocardial infarction and coronary deaths in the World Health Organization, circulation 90:583, 1994
- Jacc 2003: 41:895
10.Circ 2000, 102:2031
11.Elliott M Antman, Braunwald E Heart Disease, 7thedition. ST-Elevation Myocardial Infarction: Pathology, Pathophysiology, Clinical Features and Management
Web References
Fenton, D. E, MD. Acute Coronary Syndrome e Medicine
References for the Patient
Patient should be provided with web address where they can find additional information about heart disease:
National Library of Medicine
(www.nlm.nih.gov/medlineplus/healthtopics.html)
National Heart, Lung, and Blood Institute
American Heart Association
MCQ Questions
- Acute coronary syndrome (ACS) is the term used to describe any one of the following entities:
- stable angina
- unstable angina
- non-ST elevation myocardial infarction (non Q wave MI)
- ST elevation myocardial infarction (Q wave MI)
- stable angina
- all of the above
- a, b and c
- b, c and d
correct answer: h
- The pathophysiological substrate of an acute coronary syndrome is:
- fissure or rupture of a vulnerable plaque
- coronary spasm
- platelets activation
- thrombus formation
- all of the above
- a, b and c
- a, c and d
- a, b and d
correct answer: g
- The hallmark difference between the diagnostic of unstable angina versus non ST elevationmyocardial infarction is represented by:
- symptoms of ischemic chest pain
- EKG suggestive of ischemia
- absence of myocardial markers of necrosis (cardiac enzymes) in unstable angina
- presence of myocardial markers of necrosis (cardiac enzymes) in unstable angina
correct answer: c
- Match the coronary artery with the teritory of the left ventricle they supply:
- left anterior descending
- right coronary artery
- left circumflex coronary artery
- diagonal arteries
- septal branches of the left anterior descending artery
- posterior descending artery
- posterior wallof left ventricle
- anterior wall of left ventricle
- inferior wall of left ventricle
- anterior aspect of interventricular septum
- lateral wall of left ventricle
- posterior aspect of interventricular septum
correct option: a 2, b3, c1, d5, e 4, f6
- Occlusion of which vessel can be associated with sinus bradycardia or AV node conduction abnormalities:
- left anterior descending
- right coronary artery
- left circumflex artery
- left main
correct answer: b
- Which of the following affirmation regarding family history of cardiovascular disease is correct:
- having arelative male with MI before age 55 predisposes a person to higher risk for cardiac disease
- having a relative male with MI before age 65 predisposes a person to higher risk for cardiac
- having a relative female with MI before age 55 predisposes a person to higher risk for cardiac
- having a relative female with MI before age 65 predisposes a person to higher risk for cardiac
- a and c
- a and d
- b and c
correct answer: f
- Which of the following symptoms are suggestive of an acute coronary syndrome:
- chest pain with activity, lasting less then 30 minutes and improved by sublingual nitroglycerin
- chest pain at rest, lasting longer then 30 minutes, not responding to sublingual nitroglycerin
- epigastric pain, associated with nausea and vomiting
- numbness or tingling of left arm/wrist
- shortness of breath, palpitations, profound weakness or frank syncope
- all of the above
- a,b and c
- all of the above except a
- a, c and d
correct answer: h
- Atypical symptoms of MI are more common in:
- men
- women
- patients with other commorbidities including diabetis mellitus, heart failure or stroke
- younger patients
- older patients.
- a, c and d
- b, c and e
- a, c and e
correct answer: g
- Lack of typical chest pain in the setting of an acute MI has been reported in:
- 50 % of patients with MI
- 33% of patients with MI
c.10% of patients with MI
- 5 % of patients with MI
correct answer: b
- Which of the following conditions should be considered in the differential diagnostic of an acute coronary syndrome:
- pericarditis
- acute cholecystitis
- aortic dissection
- esophageal spasm
- pulmonary emboli or pulmonary infarct
- all of the above
correct answer: f
- Physical examination of a patient with acute MI can reveal any of the following:
- hypertension and tachycardia
- hypotension and bradycardia
- new systolic murmur or a third cardiac sound
- lung rales and jugular vein distension
- all of the above
correct answer: e
- According to World health Organization, diagnostic of acute coronary syndrome requires at least 2 of the following:
- ischemic type of chest pain
- serial EKG changes
- rise and fall in cardiac markers
- abnormal CXR
- abnormal echocardiogram
- abnormal chest CT
- a, b and e
- a, b and c
- b. d and e
correct answer: h
- EKG changes of an acute coronary syndrome are:
- ST segment depression in the leads corresponding to a territory of a coronary artery
- ST elevations more than 1 mm in at least 2 contiguous leads
- new left bundle branch block
- hyper acute T wave ( acute elevation of T wave)
- all of the above
correct answer: e
- Please match the location of the MI with the respective EKG changes:
- anterior MI
- inferior MI
- posterior MI
- lateral MI
- right ventricle
- lead II, III, aVF
- leads V2-V4
- leads I, aVL, V5 and V6
- right V4 elevation of 1 mm
- tall R in V1-V3 and ST segment depression
correct answer: a2, b1, c5,d3.f4
- The most specific enzyme for diagnostic of MI is:
- creatine kinase
b.creatine kinase MB
- troponin
d.myoglobin
correct answer: c
- What other cardiac diseases can be associated with elevation of cardiac enzymes:
a .myopericarditis
- pulmonary embolism
- aortic dissection involving the origin of coronary arteries
- cardiac trauma
- all of the above
correct answer: e
- Which of the following statements are correct:
- total creatine kinase is very specific for MI cardiac
- a ratio of CK to Ck-MB of 2.5 is usually indicative of myocardial source of CK-MB elevation
- troponins can be detected in blood at 4-6 hours post injury, peak at 24 hours andremain elevated for 7-10 days
- in patients with chest pain andSTEMI or new LBBB on EKG the decision for emergent reperfusion therapy should be based on detection of the cardiac enzymes on the blood
correct answer: b,c
- Which of the following statements are true:
- echocardiogram andCT angiography are part of routine diagnostic work-up for MI
- wall motion abnormalities seen on echo ina patient with chest pain without characteristic EKG changes support the diagnostic of ischemia
- echo is useful to detect acute valvular abnormalities secondary to ischemia or other mechanical complications
- CT angiography can rule out pulmonary emboli or aortic dissection
- all of the above
- a, b, c
- a, c , d
- b.c,d
correct answer: h
- Which of the following risk factors are part of the TIMI risk score:
- male sex, age 75, risks factors for coronary artery disease, prior aspirin use in last 7 days
- female sex, age 65,ST segment elevation, positive cardiac markers
- age 65, ST depression on EKG, known history of coronary artery disease, positive cardiac markers, prior aspirin use, more then 3 risk factors for CAD, severe angina on presentation
- ongoing chest pain, lung rales on physical exam, hypotension, and EKG changes
correct answer: c
- Which of the following statements regarding management of MI is correct:
- EKG should be taken in 10 minutes since ER arrival
- pain control can be achieved with oxygen, sublingual nitroglycerin or morphine
- patients with high risk features (ongoing chest pain, signs of pump failure, EKG changes and markedly elevated enzymes) should be admitted to coronary care unit
- cardiac catheterization should be done within 90 minutes since ER arrival in patients with chest pain and STEMI or new left bundle branch block
- fibrinolysis therapy should be started in 30 minutes since ER arrival on patients with STEMI or new left bundle block if no catheterization available in the medical facility
- all of the above
correct answer:g
21.Contraindication to fibrinolysis therapy:
- aortic dissection
- any prior intracranial bleeding
- ischemic stroke or head trauma within past 2 years
- systolic blood pressure above 150 mmHg
correct answer: a, b
- Conservative management for patients with low TIMI risk score (<2) consists of:
- aspirin
- heparin (unfractioned heparin or low molecular weight heparin)
- clopidogrel
- stress test before discharge with angiography being done if the stress test is positive for ischemia
- all of the above
correct answer: e